AMINO ACIDS
● `color{violet}("Amino acids")` are `color{violet}("organic compounds")` containing an `color{violet}("amino group")` and an acidic group as substituents on the same carbon i.e., the `color{brown}("α-carbon.")`
● Hence, they are called `color{brown}("α-amino acids.")`
● They are substituted `color{violet}("methanes.")`
● There are `color{brown}("four substituent groups")` occupying the `color{violet}("four valency positions.")`
● These are `color{violet}("hydrogen, carboxyl group, amino group")` and a variable group designated as `color{violet}("R group. ")`
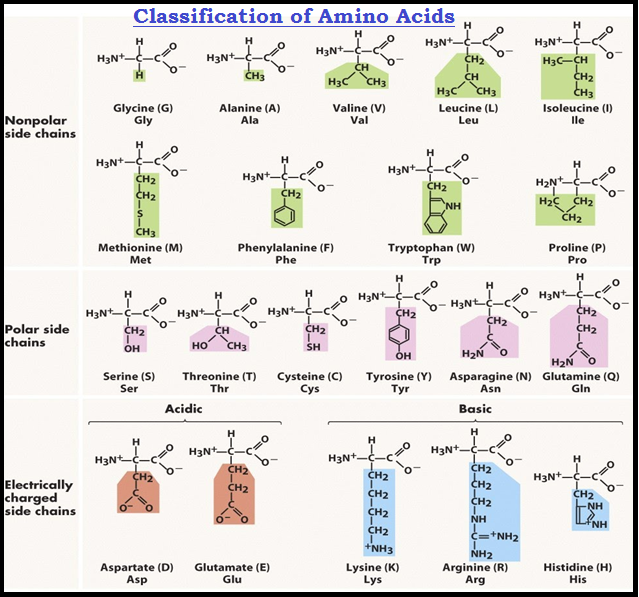
● Based on the nature of `color{violet}("R group")` there are many `color{violet}("amino acids.")`
● However, those which occur in proteins are only of twenty one types.
● The `color{brown}("R group")` in these `color{violet}("proteinaceous amino acids")` could be a `color{violet}("hydrogen")` (the amino acid is called glycine), a `color{violet}("methyl group")` (alanine), `color{violet}("hydroxyl methyl")` (serine), etc.
● The chemical and physical properties of `color{violet}("amino acids")` are essentially of the `color{violet}("amino, carboxyl")` and the R functional groups.
● Based on number of `color{violet}("amino and carboxyl groups,")` there are `color{violet}("acidic")` (e.g., glutamic acid), `color{violet}("basic")` (lysine) and `color{violet}("neutral")` (valine) `color{violet}("amino acids.")`
● Similarly, there are `color{violet}("aromatic amino acids")` (tyrosine, phenylalanine, tryptophan).
● A particular property of `color{violet}("amino acids")` is the ionizable nature of `color{violet}(–NH_2)` and `color{violet}(–COOH)` groups.
● Hence in solutions of different pHs, the structure of `color{violet}("amino acids changes.")`
● B is called `color{brown}("zwitterionic form.")`

● Hence, they are called `color{brown}("α-amino acids.")`
● They are substituted `color{violet}("methanes.")`
● There are `color{brown}("four substituent groups")` occupying the `color{violet}("four valency positions.")`
● These are `color{violet}("hydrogen, carboxyl group, amino group")` and a variable group designated as `color{violet}("R group. ")`
● Based on the nature of `color{violet}("R group")` there are many `color{violet}("amino acids.")`
● However, those which occur in proteins are only of twenty one types.
● The `color{brown}("R group")` in these `color{violet}("proteinaceous amino acids")` could be a `color{violet}("hydrogen")` (the amino acid is called glycine), a `color{violet}("methyl group")` (alanine), `color{violet}("hydroxyl methyl")` (serine), etc.
● The chemical and physical properties of `color{violet}("amino acids")` are essentially of the `color{violet}("amino, carboxyl")` and the R functional groups.
● Based on number of `color{violet}("amino and carboxyl groups,")` there are `color{violet}("acidic")` (e.g., glutamic acid), `color{violet}("basic")` (lysine) and `color{violet}("neutral")` (valine) `color{violet}("amino acids.")`
● Similarly, there are `color{violet}("aromatic amino acids")` (tyrosine, phenylalanine, tryptophan).
● A particular property of `color{violet}("amino acids")` is the ionizable nature of `color{violet}(–NH_2)` and `color{violet}(–COOH)` groups.
● Hence in solutions of different pHs, the structure of `color{violet}("amino acids changes.")`
● B is called `color{brown}("zwitterionic form.")`